Pioneering in vitro Diagnostics to Improve Public Health Worldwide
- Patient Condition
- Specialty
- Products
Patient Condition
Specialty
Product range
Products
Filter
-
RAL® STAINER
Secures Your AFB Staining and Eases Slide Reading
Easier and faster reading and no daily maintenance with RAL® STAINER. This closed fully automated bath system offers a unique fixative solution that prevents any cross-contamination.
-
RAPIDEC® CARBA NP
Rapid, Reliable Test for Carbapenemase-Producing Bacteria
This test gives reliable results in under 2 hours, making it the quick and easy way to control carbapenemase producers and meet diagnostic and screening challenges, improve patient management, and control healthcare associated infections. -
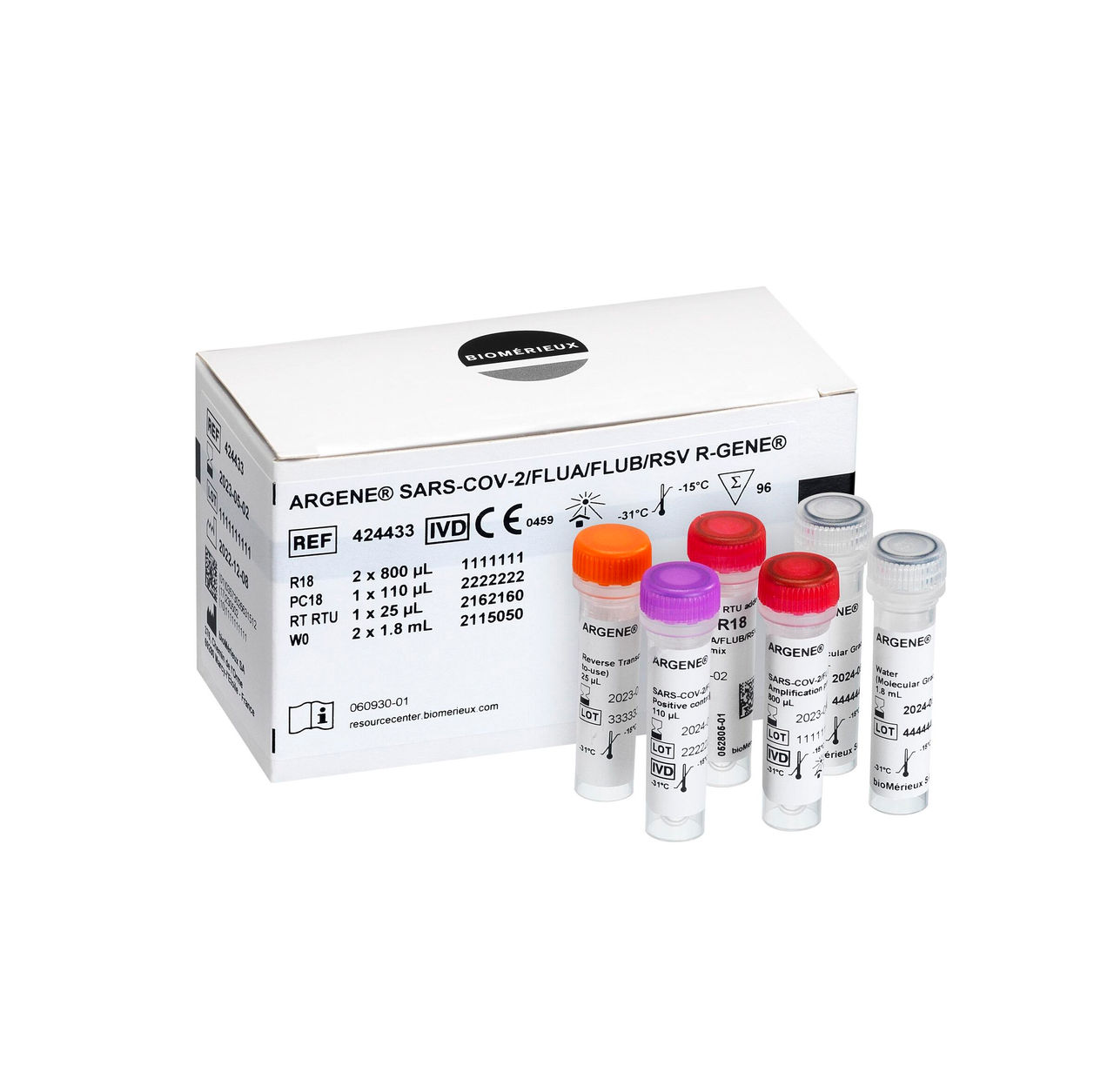
SARS-COV-2 FLUA/FLUB/RSV R-GENE®
Real-time PCR detection kit
Detect & differentiate SARS-CoV-2, Influenza A, Influenza B and RSV in 1 PCR test.
-
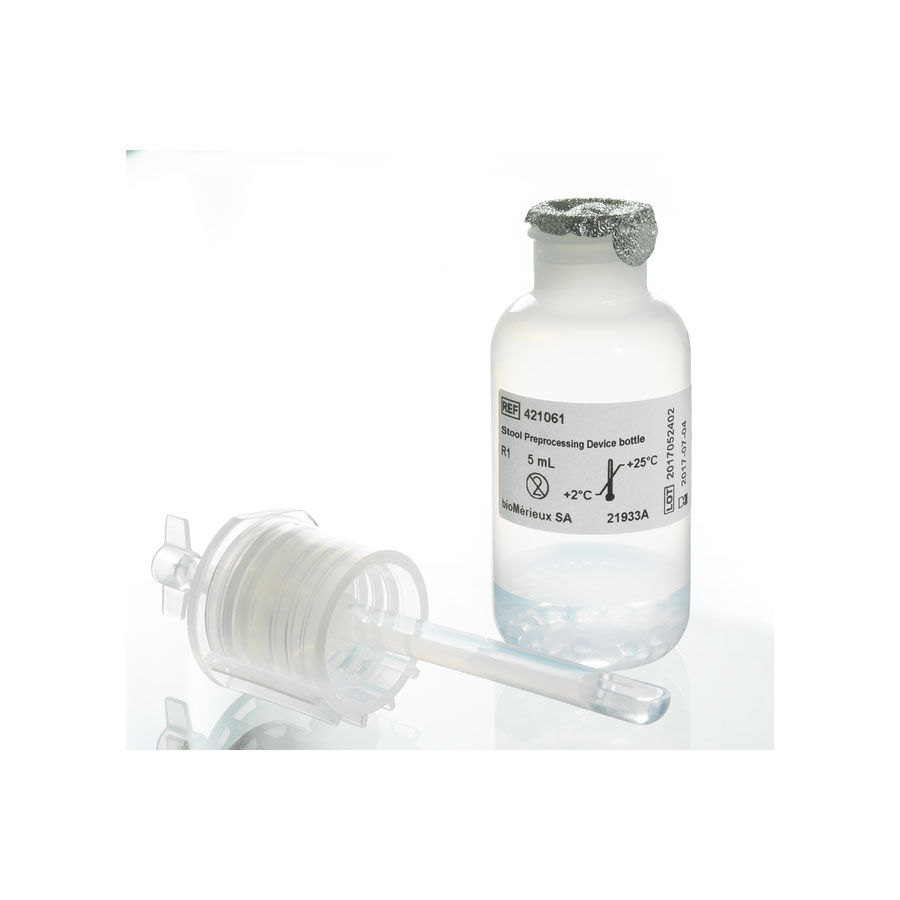
STOOL PREPROCESSING DEVICE
Stool preprocessing for molecular testing
Stool preprocessing device enabling standardized stool sample preparation for nucleic acid extraction of viruses, bacteria and parasites in about 5 minutes.
-
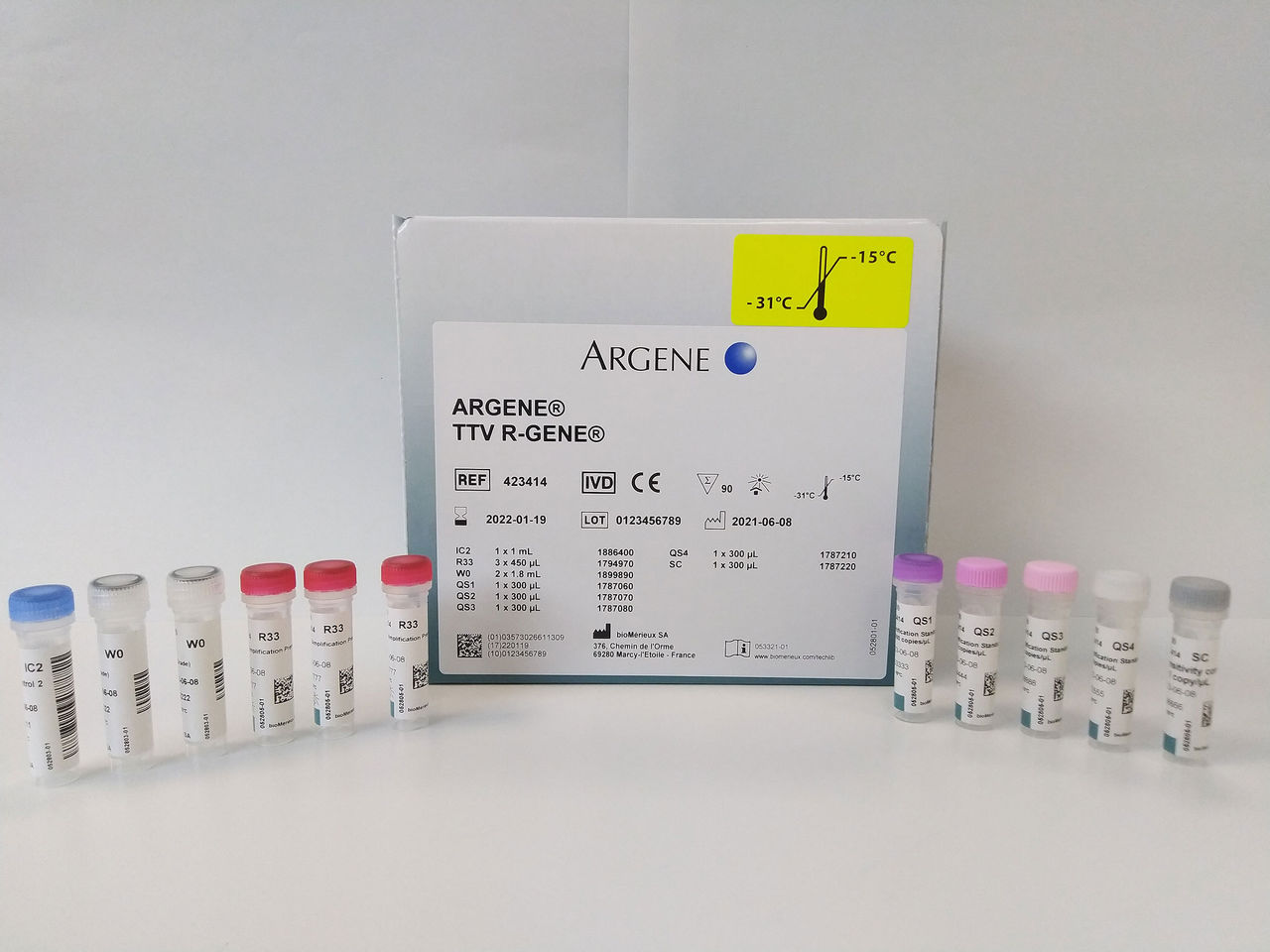
TTV R-GENE®
Guiding Transplant Risk Management
Real-time PCR assay for Torque Teno Virus detection and quantification to support transplant management.
-
VIDAS® 3
Trusted. Robust. Innovative.
With its advanced immunoassay technology and optimized testing protocols, the fully automated VIDAS® 3 benchtop immunoanalyzer empowers your lab with confidence, offering the precise results you need, when you need them.
-
VIDAS® Arboviruses Panel
Suspecting an Arbovirus? Think Dengue and Chikungunya
Our VIDAS® offer of immunoassays directed against arboviruses diseases improve access to medically important automated diagnostic tests.
-
VIDAS® BONE & MINERAL METABOLISM
VIDAS® BONE & MINERAL METABOLISM is a complete solution for the diagnosis of chronic kidney diseases, the determination of vitamin D, and iron deficiency.
-
VIDAS® C. difficile Panel
Rapid identification of C. difficile infection
The VIDAS® C. difficile GDH and TOXIN A & B (CDAB) are two complementary tests used as an aid in the diagnosis of C. difficile associated disease. NEW IVDR: VIDAS® GDH and VIDAS® CDAB are already CE Marked under the new In Vitro Diagnostic Medical Devices Regulation (EU) 2017/746 (IVDR).
-
VIDAS® Cardiac Biomarkers
Improve Patient Outcomes and Optimize Patient Management in the Emergency Department
An easy access to innovative tests for acute and complex situations to deliver reliable, fast, and actionable results to clinicians. bioMérieux with VIDAS® is providing a comprehensive emergency & critical care offer for better patient management in the emergency department.