Industry Products
-
D-COUNT® Rapid Microbial Detection
Ultra Rapid Microbial Detection
The D-COUNT® system is an advanced analysis solution for the rapid detection of microorganisms. -
CONNECT-UP™
Data Management Solution
CONNECT-UP™ is our laboratory data management solution that simplifies workflow by connecting every step—from sample to decision. -
Culture Media
Comprehensive Control
Reliable culture media solutions safeguard food products from contamination, making them an integral part of your laboratory’s daily quality control testing. -
CULTURE MEDIA For Sterility Testing
BRING RELIABILITY TO YOUR RELEASE CONTROLS
Detecting contaminants is a critical step for the release of sterile pharmaceuticals. Effective and reliable culture media will allow for the detection of any possible contaminant in your products, ensuring patient safety. -
DILUMAT®
Automated Sample Dilution
The DILUMAT® dilution system is designed to ensure accurate dilutions and reduce contamination risks, standardizing your sample preparation and evolving with your needs. -
ENDONEXT™
The Evolution of Endotoxin Testing
ENDONEXT™ endotoxin detection assays are ushering in a new era of smarter, more sustainable pharmaceutical quality control. -
ENDOXPERTS™
Comprehensive Endotoxin Services with a Special Focus on Low Endotoxin Recovery (LER)
ENDOXPERTS™ Endotoxin Services by bioMérieux provide routine testing and feasibility studies as well as specialized services for analytical issues such as LER. Our work for leading pharmaceutical companies has resulted in validated methods for LER, fulfilling regulatory requirements. -
VAXARRAY®
Designed To Reduce Your Analytical Development Bottlenecks.
VaxArray® is a multiplexed immunoassay technology for vaccine characterization. It allows manufacturers to have clearer insights into the evolution, manufacturing and release of their vaccine products.
-
ENDOZYME® II GO STRIPS
Flexible Endotoxin Testing
Laboratories don’t always need to test all 20 samples to fill a full plate—that’s why ENDOZYME® II GO STRIPS are designed with flexibility in mind: the flexibility to use only what you need, when you need it. -
ENDOZYME® II GO
Endotoxin Testing Made Faster, Easier, and Sustainable
By significantly reducing hands-on time, ENDOZYME® II GO not only makes your lab more efficient — it reduces the risk of invalid results and reduces laboratory waste by eliminating manual preparation steps that are prone to handling errors.
-
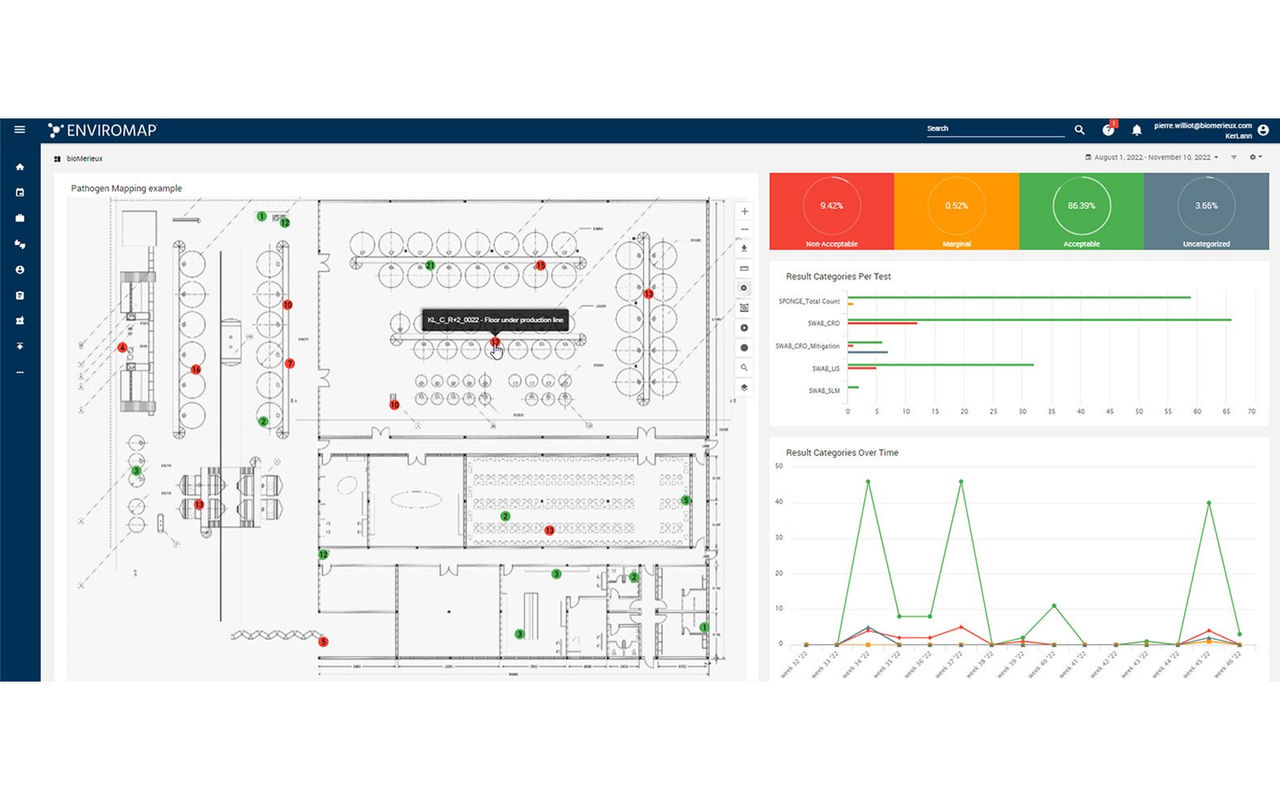
ENVIROMAP® Automated Environmental Monitoring
Elevate Your Environmental Monitoring Program
ENVIROMAP® is a secure, cloud-based system that allows you to automate your environmental monitoring program and assists with the entire sampling lifecycle.
-
ETEST®
Improving Therapeutic Decisions
The ETEST® method provides a level of precision that other manual Antibiotic Susceptibility Testing (AST) methods cannot. -
Pathogen Environmental Monitoring E-Learning Course
Elevate your team's expertise and ensure consistent planning and execution across your organization with our interactive eLearning course.
-
Feasibility Study
Quickly Confirm Your Product Compatibility and Method
We’ll prove the compatibility of your matrices with a validated bioMérieux platform depending on your microbiological specifications. -
GENE-UP®
Automated Pathogen Detection
Our real-time PCR solution simplifies workflows, decreases cross-contamination risks, and gives you rapid results that speed up critical manufacturing decisions.