Industry Products
-
CULTURE MEDIA For Sterility Testing
BRING RELIABILITY TO YOUR RELEASE CONTROLS
Detecting contaminants is a critical step for the release of sterile pharmaceuticals. Effective and reliable culture media will allow for the detection of any possible contaminant in your products, ensuring patient safety. -
D-COUNT® Rapid Microbial Detection
Ultra Rapid Microbial Detection
The D-COUNT® system is an advanced analysis solution for the rapid detection of microorganisms. -
DILUMAT®
Automated Sample Dilution
The DILUMAT® dilution system is designed to ensure accurate dilutions and reduce contamination risks, standardizing your sample preparation and evolving with your needs. -
ENDONEXT™
Better for your lab. Better for the planet.
ENDONEXT™ endotoxin detection assays are ushering in a new era of smarter, more sustainable pharmaceutical quality control. -
ENDOXPERTS™
Low Endotoxin Recovery
The phenomenon of Low Endotoxin Recovery (LER) has gained renewed attention in the scientific community in recent years as the FDA now requires that all new Biological License Applications are able to successfully unmask when LER occurs. ENDOXPERTS™ is our solution for unmasking LER, which helps you meet regulatory requirements. -
ENDOZYME® II GO
Recombinant factor C (rFC) Endotoxin detection assay
Decreasing handling factor to improve workflow efficiency, and enable faster, easier and more consistent endotoxin testing.
By significantly reducing hands-on time, ENDOZYME® II GO not only makes your lab more efficient—it reduces the risk of invalid results and reduces laboratory waste by eliminating manual preparation steps that are prone to handling errors.
-
ENDOZYME® II GO STRIPS
Flexible Endotoxin Testing
Laboratories don’t always need to test all 20 samples to fill a full plate—that’s why ENDOZYME® II GO STRIPS are designed with flexibility in mind: the flexibility to use only what you need, when you need it -
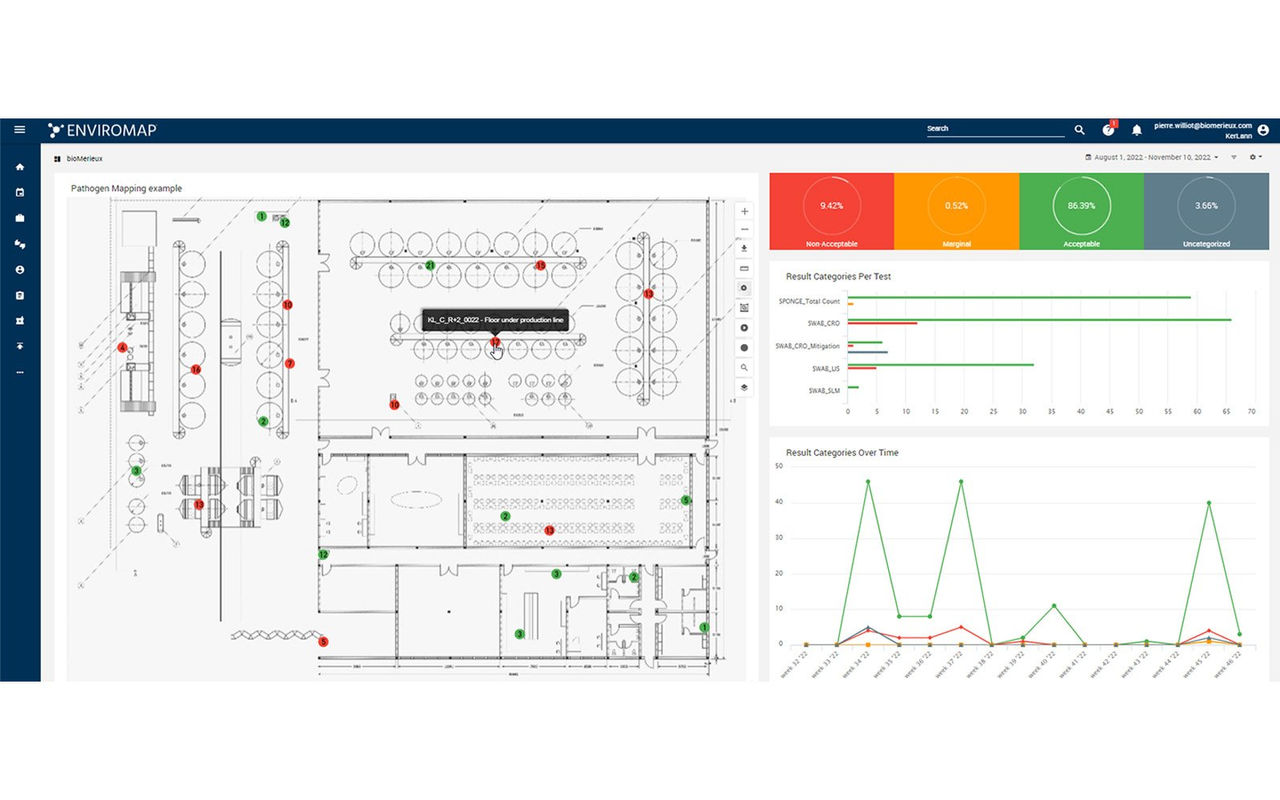
ENVIROMAP® Automated Environmental Monitoring
Elevate Your Environmental Monitoring Program
ENVIROMAP® is a secure, cloud-based system that allows you to automate your environmental monitoring program and assists with the entire sampling lifecycle.
-
Feasibility Study
Quickly Confirm Your Product Compatibility and Method
We’ll prove the compatibility of your matrices with a validated bioMérieux platform depending on your microbiological specifications. -
GENE-UP®
Automated Pathogen Detection
Our real-time PCR solution simplifies workflows, decreases cross-contamination risks, and gives you rapid results that speed up critical manufacturing decisions. -
GENE-UP® BREWPRO™ Spoilage Assessment System
Guarantee Operational Efficiency
GENE-UP® BREWPRO™ is an easy-to-use spoilage assessment system that improves overall productivity and ensures beer is clean prior to shipment well in advance of traditional microbiology assays. -
GENE-UP® CAMPYLOBACTER
GENE-UP® CAMPYLOBACTER is a real-time PCR-based solution that delivers results in under an hour and is AOAC validated for a variety of enrichment medias.
-
GENE-UP® NUTRAPLEX™ PRO microbial testing for nutraceuticals
A Paradigm Shift in Microbial Quality Testing for Nutraceuticals
Nutraceutical products are inherently complex, making recovery and detection of microbial contaminants a challenge. The streamlined GENE-UP® NUTRAPLEX™ PRO assay enables faster operations, compared to traditional methods that are time consuming and labor intensive. -
GENE-UP® PRO ACB - Alicyclobacillus spp. Detection
Alicyclobacillus spp. and Guaiacol Detection
Ensure your process control from ingredients to the bottle with GENE-UP® PRO ACB, specifically for juice quality and safety programs. -
GENE-UP® PRO BEVERAGE
Tailored Spoilage Analysis
Ensure the highest-quality customer experience with the GENE-UP® PRO BEVERAGE assay, specifically developed for the beverage industry to target the bacteria and yeasts responsible for spoilage.